USFDA Recall: Nutramigen
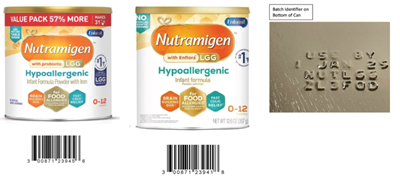
Today, the U.S. Food and Drug Administration (FDA) is alerting parents, caregivers, and health care providers that Reckitt/Mead Johnson Nutrition has voluntarily recalled certain Nutramigen Hypoallergenic Infant Formula Powder products due to possible Cronobacter sakazakii contamination. No illnesses have been reported to date in connection with this recall, and it is likely most of the product that was distributed in the U.S. has already been consumed.


